
For example, the correct name for the compound shown below is 3‑methylheptane, not 2‑ethylhexane. 
When naming alkanes, a common error of beginning students is a failure to pick out the longest carbon chain. Perhaps the name you should have written was 3‑ethyl-4,4‑dimethylheptane! If you were to check the given structure and find 11 carbon atoms, you would know that you had made a mistake. For example, if you named a compound 3‑ethyl-4‑methylheptane, you have indicated that the compound contains a total of 10 carbon atoms-seven carbon atoms in the main chain, two carbon atoms in an ethyl group, and one carbon atom in a methyl group. 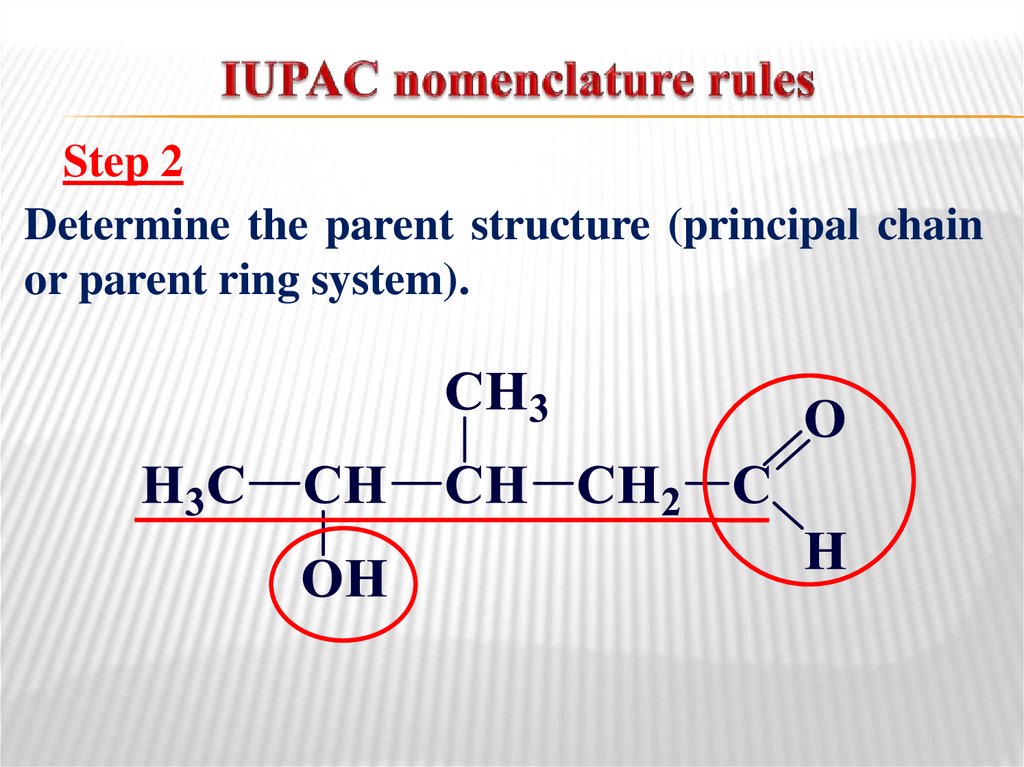
One way of checking whether the name you have given to an alkane is reasonable is to count the number of carbon atoms implied by the chosen name.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
